Abstract
Background
The three Food and Drug Administration (FDA)-approved anti-tumor necrosis factor drugs (anti-TNFs) for Crohn’s disease (CD) have not been directly compared.
Aim
To compare the efficacy of the three anti-TNFs for CD in clinical practice.
Methods
Retrospective review of patients initiated on anti-TNF between 2004 and 2008. Disease activity, quality of life, and remission rates were compared between groups over 1 year.
Results
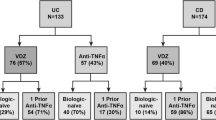
Sixty patients with CD were initiated on anti-TNF from 2004 to 2008: 31 on infliximab (IFX) and 29 on adalimumab (ADA) or certolizumab pegol (CTZ). More patients in the ADA/CTZ scores group had prior exposure to anti-TNF (76 versus 10 %, p < 0.01). Mean Harvey–Bradshaw Index (HBI) scores in the IFX group were lower than in the ADA/CTZ group at 12 months (2.72 ± 3.34 versus 5.63 ± 5.33, p = 0.03). At 12 months, more IFX patients were in remission compared with those on ADA/CTZ (88 versus 53 %, p ≤ 0.01). Mean Short Inflammatory Bowel Disease Questionnaire (SIBDQ) scores were not different between the IFX and ADA/CTZ groups at 12 months. Stratified analyses and logistic regression based on prior anti-TNF use did not show differences in remission rates at any time point post-baseline between groups.
Conclusions
After adjustment for prior anti-TNF there was no difference in remission rates between the IFX and ADA/CTZ groups at any time point post-baseline. This suggests that differences between groups were accounted for by a higher rate of prior anti-TNF in the ADA/CTZ group. Our results should be reviewed with caution given the small sample size.




Similar content being viewed by others
Abbreviations
- CD:
-
Crohn’s disease
- IBD:
-
Inflammatory bowel disease
- EIM:
-
Extraintestinal manifestations
- IFX:
-
Infliximab
- ADA:
-
Adalimumab
- CTZ:
-
Certolizumab pegol
- HBI:
-
Harvey–Bradshaw Index
- SIBDQ:
-
Short Inflammatory Bowel Disease Questionnaire
- CDAI:
-
Crohn’s Disease Activity Index
- IBDQ:
-
Inflammatory Bowel Disease Questionnaire
- IM:
-
Immunomodulator
References
Van Deventer SJH. Tumor necrosis factor and Crohn’s disease. Gut. 1997;40:443–448.
Targan SR, Hanauer SB, van Deventer SJ, et al. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn’s disease. Crohns disease cA2 study group. N Engl J Med. 1997;337:1029–1035.
Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn’s disease: the ACCENT I randomised trial. Lancet. 2002;359:1541–1549.
Present DH, Rutgeerts P, Targan S, et al. Infliximab for the treatment of fistulas in patients with Crohn’s disease. N Engl J Med. 1999;340:1398–1405.
Sands BE, Anderson FH, Bernstein CN, et al. Infliximab maintenance therapy for fistulizing Crohn’s disease. N Engl J Med. 2004;350:876–885.
Hanauer SB, Sandborn WJ, Rutgeerts P, et al. Human anti-tumor necrosis factor monoclonal antibody (adalimumab) in Crohn’s disease: the CLASSIC-I trial. Gastroenterology. 2006;130:323–333.
Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn’s disease: the CHARM trial. Gastroenterology. 2007;132:52–65.
Sandborn WJ, Rutgeerts P, Enns R, et al. Adalimumab induction therapy for Crohn’s disease previously treated with infliximab: a randomized trial. Ann Intern Med. 2007;146:829–838.
Nesbitt A, Fossatti G, Bergin M, et al. Mechanism of action of certolizumab pegol (CDP870): in vitro comparison with other anti tumor necrosis factor alpha agents. Inflamm Bowel Dis. 2007;13:1323–1332.
Sandborn WJ, Feagan BG, Stoinov S, et al. Certolizumab pegol for the treatment of Crohn’s disease. N Engl J Med. 2007;357:228–238.
Schreiber S, Khaliq-Kareemi M, Lawrance IC, et al. Maintenance therapy with certolizumab pegol for Crohn’s disease. N Engl J Med. 2007;357:239–250.
Lichtenstein G, Thomsen O, et al. Long-term remission with Certolizumab Pegol in Crohn’s disease over 3.5 Years: results from the PRECiSE 3 study. Am J Gastroenterol. 2009;104:S450.
Baert F, Noman M, Vermeire S, et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn’s disease. N Engl J Med. 2003;348:601–608.
Karmiris K, Paintaud G, Noman M, et al. Influence of trough serum levels and immunogenicity on long-term outcome of adalimumab therapy in Crohn’s disease. Gastroenterology. 2009;137:1628–1640.
Satsangi J, Silverberg MS, Vermeire S, et al. The Montreal classification of inflammatory bowel diseases: controversies, consensus, and implications. Gut. 2006;55:749–753.
Vermeire S, Schreiber S, Sandborn WJ, et al. Correlation between the Crohn’s disease activity and Harvey-Bradshaw indices in assessing Crohn’s disease severity. Clin Gastroenterol Hepatol. 2010;8:357–363.
Jowett SL, Seal CJ, Barton JR, et al. The short inflammatory bowel disease questionnaire is reliable and responsive to clinically important change in ulcerative colitis. Am J Gastroenterol. 2001;96:2921–2928.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patil, S.A., Rustgi, A., Langenberg, P. et al. Comparative Effectiveness of Anti-TNF Agents for Crohn’s Disease in a Tertiary Referral IBD Practice. Dig Dis Sci 58, 209–215 (2013). https://doi.org/10.1007/s10620-012-2323-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2323-0